
The FDA went around the usual processes and regulatory channels and signed off on the test under an Emergency Use Authorization, allowing the use of detection kits...
Read More
Based in Atlanta, BP Genomics has launched a molecular test that detects only the 2019 strain of the coronavirus. They have collaborated with a number of partners to develop a kit that extracts the RN...
Read More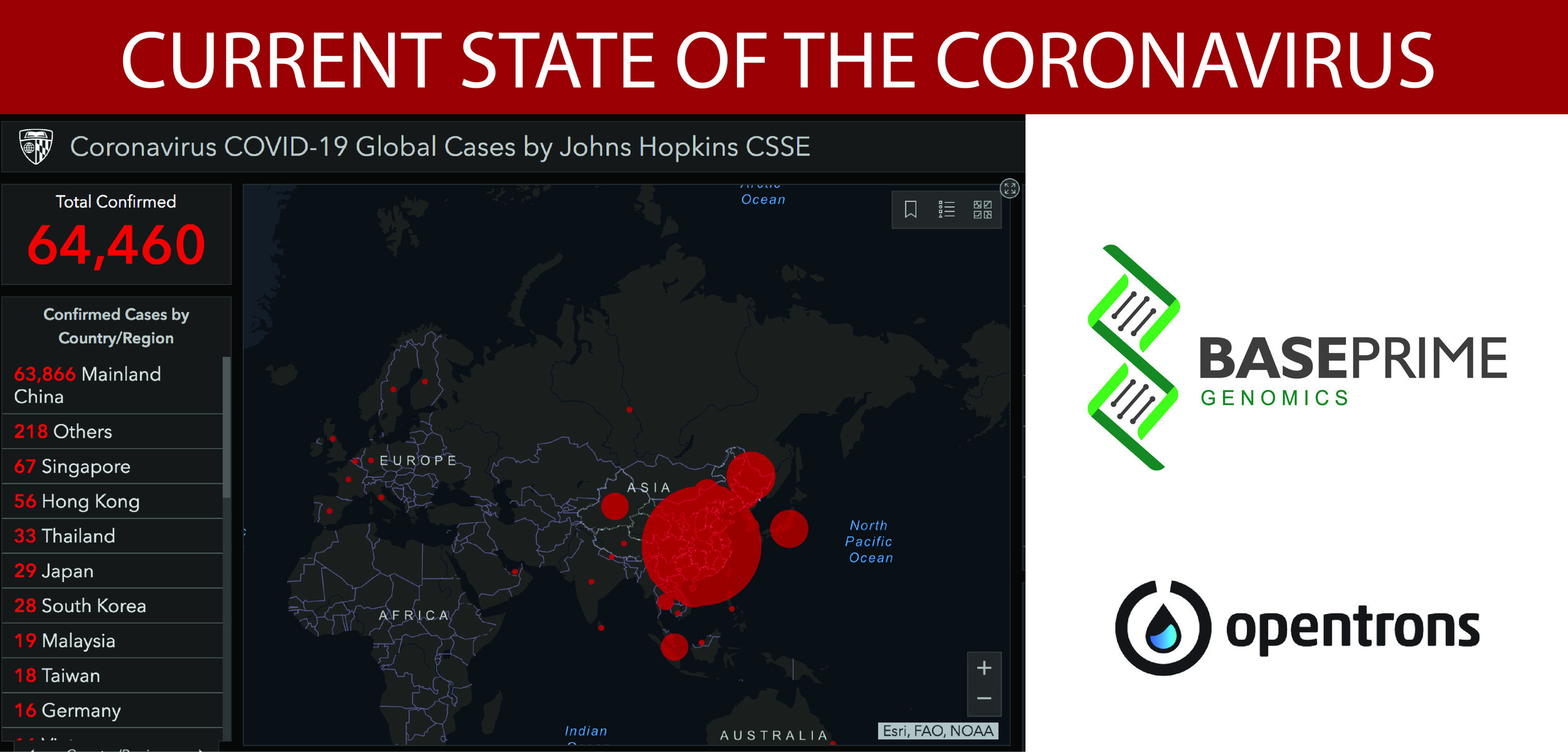
BP Genomics, an innovative leader in detection assays, sample processing, and nucleic acid technologies has partnered with Opentrons, a unique and forward thinking liquid handling company, whose passi...
Read More